/PeriodicTable-58b5d9253df78cdcd8d03ebb.jpg)
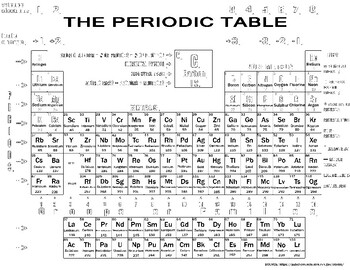
Hint: Count the number of atoms of each element, and then multiply that number by the element's atomic weight. Use the table below to find the atomic weight of each atom (element), or refer to a Periodic Table of the Elements. Let us calculate the molecular weight of some common compounds. For example, in one mole of a chemical compound there are 6.022 x 1023 molecules. Solution: Use only two digits after the decimal point, making the value that would be used in calculations (35.45). Round this to the correct number of digits. Example 2: One periodic table lists the molar mass of Chlorine to be (35.453) grams per mole. One mole of 'something' contains 6.022 x 1023 entities. As a rule of thumb, always use the value rounded to two digits after the decimal point. A mole is the unit that measures the amount of a substance. One thousand mers connected together would add up to a weight of 28,000 grams/mole and would have 6,000 atoms.Ī mole is the standard method in chemistry for communicating how much of a substance is present. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. We combine (react) many mers of ethylene together to form a polyethylene chain. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.The chemical symbol for Hydrogen is H. The atomic weight of carbon is 12 and that of hydrogen is 1, so one mer of ethylene has a weight of 2(12) + 4(1) = 28. The given mass of K (4.7 g) is a bit more than one-tenth the molar mass (39.10 g), so a reasonable ballpark estimate of the number of moles would be slightly greater than 0.1 mol. It has a total of 6 atoms: 2 carbon (C) atoms and 4 hydrogen (H) atoms. Referring to the periodic table, the atomic mass of K is 39.10 amu, and so its molar mass is 39.10 g/mol. The chemical formula for an ethylene monomer is -(CH2-CH2).Every hydrogen atom has one proton in its nucleus. This color periodic table contains the accepted standard atomic weights (atomic masses) of each element as accepted by the IUPAC. Hydrogen, at the upper left of the table, has an atomic number of 1. In this table, an elements atomic number is indicated above the elemental symbol. In order to calculate the molecular weight of one water molecule, we add the contributions from each atom that is, 2(1) + 1(16) = 18 grams/mole. The periodic table (see figure below) displays all of the known elements and is arranged in order of increasing atomic number. Using the periodic table of the elements to find atomic weights, we find that hydrogen has an atomic weight of 1, and oxygen's is 16. The chemical formula for water is H2O, which means this molecule has 3 atoms: 2 of hydrogen (H) and 1 oxygen (O) atom.
